Background
Relapse in patients with acute myeloid leukemia (AML) has been, in part, attributed to the ability of leukemic blasts to escape immune surveillance through mechanisms such as MHC class II antigen modulation, alteration of the cytokine milieu, and upregulation of inhibitory and immune checkpoint ligands that promote T-cell suppression. Yet, immune checkpoint inhibitors (ICI) used in AML have yielded variable and unpredictable results. Moreover, evaluation of T-cell function is not part of the diagnostic assessment of patients with AML.
Methods
To better characterize the T-cell dysfunction in AML, we conducted a prospective study on freshly harvested, bone marrow aspirates from patients with AML at presentation, collected under an Institutional IRB-approved banking protocol over the past two years. Using a multiparameter flow cytometry-based assay, we analyzed T-cell activation potential in response to CD3/CD28 (bead-based), T-cell receptor (TCR) stimulation, both alone (n= 40 AMLs and 10 healthy donors) or in combination with a library of ICI (n=15 AML and 5 healthy donors). We selected OX40 (CD134/TNFRSF4) and ICOS (Inducible T-cell costimulator) as surrogate markers for T-cell activation based on their expression pattern post bead-based stimulation in healthy donors' bone marrows. The ICI library comprised 31 therapeutic antibody inhibitors of known immune checkpoints, signaling pathways, and chemokines reported to be implicated in T-cell suppression in AML and other solid tumors.
Results
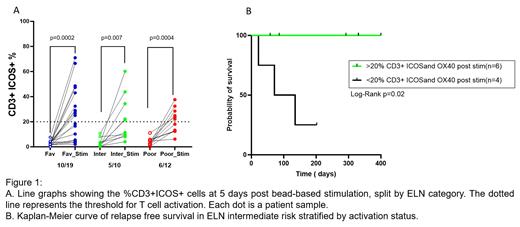
Healthy T-cells displayed the highest surface expression of OX40 and ICOS at 5 days post-bead-stimulation, which was selected as the endpoint for our in vitro analyses. We found that bone marrow-derived T-cells from healthy donors upregulated the expression of the markers ICOS and/or OX40 in at least 20% of T-cells post-stimulation (n= 10, ICOS: median 36.9%, range 20-72%; OX40: median 59.3%, range 21.4 -83%). Overall, AML samples displayed a lower degree of ICOS and OX40 T-cell expression in response to TCR stimulation (n=40, ICOS: median 20.5%, range 4-65%; OX-40: median 30.3%, range 0.3-80%). Based on this result, T-cell activation was defined as >20% CD3+ cells ICOS+/OX-40+ with stimulation, with greater than 2-fold changes from baseline ICOS and OX40 expression. Using this criterion, patients with AML were then classified as activators or non-activators. T-cell activation status correlated with ELN 2022 risk (1) (Figure 1A). For the intermediate risk group, we observed that the activators group had a significantly longer relapse-free survival when compared to the non-activators (Figure 1B, n=10, Mantel-Cox log-rank = 0.02, follow up 400 days at the time of writing). While a similar trend was observed in the favorable and the poor risk groups, it did not reach significance in these ELN categories. To assess if the T-cell activation could be rescued or augmented upon immune checkpoint inhibition, we tested the effect of each of the 31 compounds of the ICI library on the degree of T-cell activation in diagnosis bone marrow aspirates of 15 patients (7 activators and 8 non activators) and in 5 healthy donors. Matched peripheral blood (PB) samples were available for 5 of the 15 patients with AML. All tested normal donors reached the threshold for T-cell activation, which was not significantly affected by ICI treatment. The 7 AML activators behaved similarly to the healthy donors. In 2 of the 8 AML non-activators, selected ICI rescued T-cell activation in absence of TCR stimulation, while in the remaining 6 cases, T-cell activation was not achieved under any condition. Similar, but overall dampened activation trends, were observed in PB samples.
Conclusions
Consistent with the findings of our previous retrospective study (2), the ongoing prospective analysis of T-cell activation of diagnosis bone marrows of patients with AML suggests that testing TCR-mediated T-cell activation potential at diagnosis may help refine risk stratification of the heterogenous ELN intermediate risk group. AML immune suppression seems to be reversible in a minority of cases with the use of selected ICI inhibitors.
References
1. Blood(2022) 140 (12): 1345-1377
2. Proc Natl Acad Sci U S A. 2021 Dec 7;118(49)
Disclosures
Cashen:Kite Pharma: Consultancy. Uy:Jazz: Other: Advisory Board.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal